Orbital diagrams show the way these electrons order themselves. Atoms with electrons organized in their orbitals are in their ground state, or their most stable energy level. To do this, the electrons within the atom occupy orbitals, specific areas in the atom, that keeps the energy low (since the energy in the atom rises the closer electrons are to each other). 
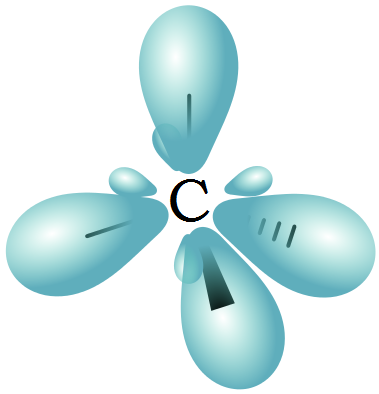
The Ground Stateīefore we get deep into how orbital diagrams help to explain hybridization, I’d like to go summarize the last two arcs in a few sentences (yes, really).Ītoms seek to lower their energy levels to be in their most stable state. To conceptually understand what hybridization looks like, we have to revisit the orbital diagram - the familiar tool that we first used when discussing Hund’s Rules. However, since hybridization goes deeper than our valence electron-based framework, we can use it to explain: (1) the electron-pair geometry and (2) what a “covalent bond” really is. But hybridization gives us another way to explain covalent bonding - as a function of overlapping orbitals. Up until now, we’ve explained bonding using valence electrons. No, it turns out that they remain as they are, but overlap with each other to create a unique hybrid of orbitals. 
Hybridizationĭo you remember orbitals? Those things we learned about that were composed of those four quantum numbers? To recap: the principle quantum number (n) represents the electron “shell”, or the energy level of any given electron, the orbital quantum number (l) further divides those shells into smaller “subshells”, the magnetic quantum number (m) tells us the number of ways that subshells can orient themselves within a shell, and the spin quantum number (s) an electron’s momentum, or “spin”.Īll four of these numbers come together to provide an understanding of paths that electrons can take within any atom, which take the form of specific shapes.īut what happens to these orbitals when atoms bond? Do they change shape? Do they disappear and form some completely different orbital? And, it turns out that this relationship, like many things you will see from here on out, is based on science that we’ve already covered. Toward the end of the last section, I alluded to a relationship between orbitals and molecular geometry. What theory (or theories) explain this behavior? So, given our understanding that atoms bond to reduce their energy, that atoms use their valence electrons to do so and, when they do so, they take different shapes, the next course of action is to backtrack. But, as always, where there is a will to explore something to its concrete conclusion, there is always an assumption that drove it. Scientists established the exact angles and geometries between atoms through observation and experimentation. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
